Hi all,
I’m relatively new to VENUS programming so excuse me if this is an easy-to-fix question. I regularly have to deal with tubes with a pellet of tissue/zirconia beads covered with ethanol. My plan is to remove as much ethanol as possible.
I already have a working solution but I think this could be further improved. I do a slow aspirate from the surface with cLLD and an aspirate all step. That removes about 80% of the ethanol until the tip reaches the tissue and clogs. Then I dispense the ethanol into the waste, wait for 20s and do another aspirate.
My idea is to monitor the pressure instead of doing an aspirate all, cause I guess that VENUS should be able to throw an error once the tip is clogged (meaning the TADM curve overshoots?) and repeat this procedure 1 time to aspirate as much as possible. The problem is: I don’t know how to implement this and I can’t find anything about it in the “normal” error handling.
Thanks for your help in advance,
Best Dominik
@dominik.b - Yes, TADM can and has been used for such instances (phase separation, sample rescue prior to clogs etc.), and IMO really showcases the capability of our channels when leveraged appropriately. Depending on the application though, there can be some nuance involved, and would need some tailoring and customization per the specific aspects and requirements of the particular sample transfer, and a forum post may not be the best venue for attempting to capture that level of detail and information for a full implementation.
For more background on TADM monitored transfers, I highly recommend checking out section 7.2 of the Liquid Handling Reference Guide linked in Eric’s shared materials folder, found in this post. Here are the cliffnotes there for the working principle:
-
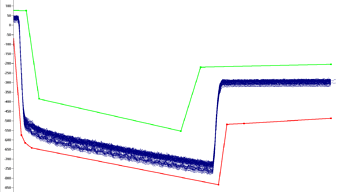
Internal pipette pressure sensors monitor and capture real-time pressure data during aspiration and dispense (ex aspiration):
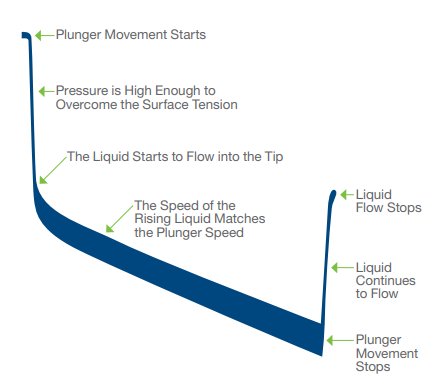
-
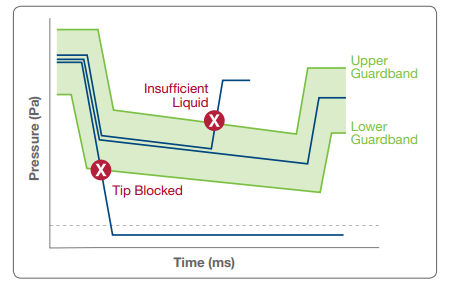
For defined transfers of a consistent volume, the liquid editor can be used to define upper and lower gaurdbands that define the allowable pressure values for a given routine transfer:
-
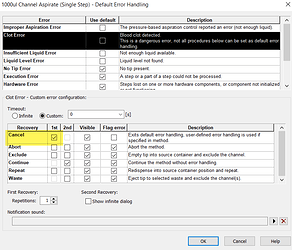
At run time, if there is a clog, during an aspiration, it will immediately trigger a significant underpressure, where the pressure curve will drop out below the allowable bottom threshold. When TADM monitoring is enabled in system configuration, this will trigger a clot error, which can be configured for custom error handling code to handle the clogs.
-
One would then parse the step data of the aspiration to see which channels/samples were affected, which is another topic entirely.
This is a pretty advanced technique, and for a new user I would highly encourage reaching out to your local applications team for support if this is your only option for this transfer. We can also put you in touch if you would like to be pointed in the right direction.
Given the relative complexity and custom nature of the above approach though, I typically only recommend it if this is your only option.
All that said, depending on your scenario it may not be required to accomplish your transfer, and there may be simpler means. Are the pellets consistent in size and surface? Would the pellet be less prone to prematurely generating a clog if you lowered the aspiration plunger speed? What is the reasoning for the 20s wait time in between aspirations?
-Nick
3 Likes
Hi Nick,
wow, thanks for that reply, this is really comprehensive. I guess my case it would be possible to define TADM curves since this is a simplified problem. The maximum volume is always 1 mL, so I could look at a TADM curve of an unclogged aspirate and just set the lower threshold as a straight line since I’m only interested in the clogging issue.
To answer your questions:
- No the pellets are not consistent in size and surface. The volume is always 1 mL but the ratio from pellet to liquid differs substantially and is sometimes even 0 for the negative controls. The pellet consists of homogenized insect tissue, so I can afford to throw a bit of it away.
- Yes, lowering the plunger speed by adjusting the liquid class greatly increased the amount of ethanol that was removed from the pellet, so the tip clogged at a later point in the aspiration.
- The idea of the pause would be to let the tissue settle for a short time after it was disturbed by the pipette tip. After that, I was able to remove another ~ 100 uL, which would then in total be sufficient I guess.
All said: It could already work like this but being a perfectionist I guess the RIGHT way to do this would be to stop aspirating once the tip clogs, dispense everything in the tip into the waste and do a 2nd / 3rd aspirate again until the clogging appears.
In the advanced course I learned how to parse the error code, but I guess in this case I could just use an error handling by user to dispense all tips into the waste?
best Dominik
2 Likes
@dominik.b - No problem, happy to help!
It sounds like your current approach may already be sufficient for practical purposes. The max removal volume of 1mL in your case also greatly simplifies things, as well as the fact that the pellets wont be lost (besides maybe a couple beads) in the event of a clog.
TADM monitoring doesn’t sound like it is required for success here, especially if you already have a working solution. Of course, you could choose to implement it if you wanted to fully optimize, but that is up to you.
Even if you opted to use TADM here, it would be a bit simpler. You wouldn’t even need to parse step data for channel specific clogs, as all channels would be dispensing to waste whether they clogged or not. You would just cancel the error and could have an empty custom error handling block and it would probably still work.
-Nick
3 Likes
Okay, I got quite far. TADM is installed, I figured how to set up the bands in the liquid class, however I struggle to get any recordings for my channels. If I want to load them from the run they don’t seem to be in the LogFiles folder.
In the trace it says that it’s uploading the TADM curves, but I can’t find them. Any hint here?
best
Dominik
Hi Dominik,
I uploaded the TADM manual - in section 4.5.1 it explains how to view the TADM curves via the Liquid Class Editor.
When TADM is enabled, every run creates an additional *.mdb file with an “_tadm” suffix in the LogFiles folder. That is what the Liquid Class Editor is looking for when you select “add from run” and it prompts you to select runs and defaults to the Logfiles directory.
-Eric
2 Likes
Thanks Eric, it’s working perfectly 
You guys cannot guess what a revolution it is for me to switch from our old FxP to the Vantage. I could program 12h per day 
4 Likes
So news on this topic:
I finished programming the method. Whenever a clog is detected, the Vantage will dispense to waste and aspirate a second time to catch some of the remaining ethanol, while discard all tips that did not clog, since there is no reason to aspirate a second time if the first aspiration was successfull. All cloggs are detected, TADM bands seem to work great.
But I encountered one last issue: When a clog is detected the tips are removed from the liquid at full speed leading to droplet formation due to remaining ethanol on the outside of the tip. I thought about having 2nd 0 volume dispense in the same sample to bypass this but it seems like a lot of additional movement, that could and should be solved in a smoother way. Any ideas on this?
best Dominik
@dominik.b - When the aspiration step contains channels that use a cancel error recovery, they will bypass swap speed from the liquid class, and will instead retract at normal Z motor speed.
Are you using liquid level detection or fixed height aspirations? Using level detection and following, combined with a minimal submerge depth should result in an essentially negligible amount of liquid surrounding the end of the tip at the time a clog is generated.
-Nick
1 Like
Hi Nick,
I use cLLD and the droplet is minimal, however since I do some handling inbetween it stays in the air for some time. However I discovered ADC and it works perfectly for this application. So my problem is fixed already. Thanks for the quick response though, I really appreciate having a community to discuss problems with 
1 Like
Ah I see. Then the droplet is actually not forming due to ethanol collecting on the outside of the tip, but rather pressure buildup in the headspace of the tip due to volatility/generation of a vapor phase. For ethanol, typically air transport volume (air gap drawn after aspiration of liquid) will prevent droplets for typical transfers.
When the step is cancelled for the channel, there is no more plunger movement, so you aren’t getting an airgap for the clotted tips.
Good job using ADC to resolve this!
-Nick